Osmotic signaling governs sunscreen biosynthesis to safeguard desert cyanobacteria against desiccation
GA, UNITED STATES, April 27, 2026 /EINPresswire.com/ -- Mycosporine-like amino acids (MAAs) are natural sunscreens synthesized by a wide range of organisms, yet their functions beyond UV protection and the associated regulatory pathways remain poorly understood. Here, the authors demonstrate that MAAs serve as essential osmoprotectants in the desiccation tolerance of the desert cyanobacterium Nostoc flagelliforme. Genetic disruption of MAA biosynthesis impairs growth, elevates oxidative damage, and compromises photosynthesis under dehydration. Moreover, MAAs are shown to stabilize proteins and scavenge reactive oxygen species. The authors also identify the Dsp1-OrrA two-component system as a specific osmotic signaling pathway that governs MAA induction. Loss of either Dsp1 or OrrA abolishes osmotic-induced MAA accumulation and severely reduces desiccation tolerance. These findings redefine MAAs as multifunctional guardians and reveal a novel regulatory axis for extremophile adaptation.
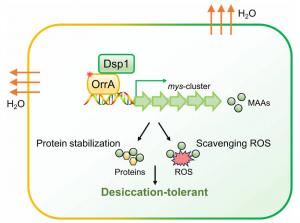
Mycosporine-like amino acids (MAAs) are natural sunscreen compounds widely distributed across bacteria, algae, and corals. While their induction by ultraviolet radiation is well established, their potential roles in other stresses and the underlying signaling pathways have remained largely unexplored. Here, the authors present compelling evidence that MAAs function as effective osmoprotectants to safeguard the desert cyanobacterium Nostoc flagelliforme against desiccation, and they identify a novel Dsp1-OrrA signaling module that governs osmotic induction of MAA biosynthesis (Fig. 1).
MAA biosynthesis in Nostoc flagelliforme is significantly induced by water-deficit stress, such as sorbitol or PEG treatment. Using CRISPR-Cpf1 gene editing, the authors generated a mysA knockout mutant that completely lost MAA production. Under sorbitol-induced water deficit stress, the mutant exhibited severe growth inhibition, photosynthetic impairment, and elevated oxidative damage compared to the wild-type strain. In vitro biochemical assays revealed that MAAs directly stabilize desiccation-sensitive proteins, such as lactate dehydrogenase (LDH), and efficiently scavenge reactive oxygen species, including superoxide anions and DPPH radicals. These results demonstrate that MAAs play dual protective roles as both osmoprotectants and antioxidants, which is essential for the desiccation tolerance of Nostoc flagelliforme.
To elucidate the regulatory mechanism, the authors investigated the transcription factor OrrA, which was previously shown to bind the mysA promoter. Deletion of orrA abolished osmotic-induced MAA accumulation and severely reduced desiccation tolerance. Notably, the expression of orrA remained constant under sorbitol-induced osmotic stress, indicating the presense of an unknown upstream dehydration-responsive regulator. Using co-immunoprecipitation and mass spectrometry, the authors identified a histidine kinase, designated Dsp1 (dehydration-sensing protein 1), that physically interacts with OrrA. Yeast two-hybrid and pull-down assays confirmed this interaction. The expression of dsp1 was significantly induced by sorbitol-induced osmotic stress, suggesting its role as an upstream dehydration-responsive regulator. In the Δdsp1 mutant, osmotic-induced MAA biosynthesis was significantly inhibited, and the strain showed marked sensitivity to dehydration, including reduced photosystem II activity and impaired recovery after desiccation-rehydration cycles. Further analysis revealed that the Dsp1-OrrA module may also participate in regulating the biosynthesis of classical osmoprotectants, including sucrose and trehalose, suggesting that it functions as a key regulatory hub that coordinates the synthesis of multiple osmoprotectants to confer desiccation tolerance in cyanobacteria. In contrast, UV-induced MAA production remained largely unaffected in both the Δdsp1 and ΔorrA mutants, indicating their dehydration-specific regulatory role.
Phylogenomic analysis revealed that the five-gene mys biosynthetic cluster is selectively conserved in desiccation-tolerant cyanobacteria, and the co-occurrence of dsp1 and orrA with this cluster supports their adaptive significance. These findings redefine MAAs as multifunctional guardians that provide both UV protection and desiccation tolerance, and uncover a novel regulatory axis underpinning stress adaptation in extremophilic microorganisms. This work establishes MAAs as key osmoprotectants in desiccation tolerance of desert cyanobacteria, and provides a foundation for engineering stress-resistant microbes and for the sustainable biotechnological production of these multifunctional compounds.
Original Source URL
https://doi.org/10.1002/mlf2.70075
Funding information
This work was supported by the National Natural Science Foundation of China (Nos. 32430005 and 32470408), the Wuhan Science and Technology Major Project (No. 2023020302020708), and the Fundamental Research Funds for the Central Universities (CCNU25JC032).
Lucy Wang
BioDesign Research
email us here
Legal Disclaimer:
EIN Presswire provides this news content "as is" without warranty of any kind. We do not accept any responsibility or liability for the accuracy, content, images, videos, licenses, completeness, legality, or reliability of the information contained in this article. If you have any complaints or copyright issues related to this article, kindly contact the author above.